Which one is more concentrated with water? The one with the fewest particles (10 teaspoons). If I have 2 identical cups with the same amount of water, and I add 20 teaspoons of lemonade to one cup and 10 teaspoons of lemonade to the other cup, which cup is more concentrated with lemonade? The one with the most particles (20 teaspoons).Water also wants to move from its area of high concentration to an area of low concentration. The next force of nature that will kick in is water diffusion.However, the cell membranes will not allow them to pass, so they are stuck inside the cell.The particles in the cells will want to move into the plasma.Therefore, since the particles in the cells are 300 mOsm and the particles in the blood vessel are 260 mOsm, which way do the particles want to move?.By a law of physics, all particles want to move from an area of high concentration to an area of low concentration…That is why perfume will diffuse out of the bottle and fill up the room if you leave the top off. If the plasma becomes diluted to 260 mOsm, and the cells next to a blood vessel are still at 300 mOsm, the cells now have more particles.0.3 Osmolal = 300 mOsmolal (actually closer to 280mOsmolal) Plasma Interstitial Intracellular Same osmolalities (total number of particles) When you evaporate away the liquid in both coffee cups and count each coffee grain and each sugar grain, there are 300 million total grains per liter in both cups.Different volumes(one coffee cup holds 8 oz, another holds 16 oz).For example, one coffee cup has 1 teaspoon of instant coffee and 2 teaspoons of sugar, another cup has 2 teaspoons of coffee and 1 teaspoon of sugar. Different compositions (different amounts of individual particles).Water always moves across the compartments because cell membranes always allow water to pass.If one compartment has more particles than another one next to it, and if those particles cannot reach equal numbers on their own because the cell membrane blocks their passage, water will try to dilute the compartment with the higher number of particles until they are at the same number of particles per liter.That means that there are 300 million particles (or 300 milliosmoles, abbreviated 300 mOsm) dissolved in each liter of water in each compartment.It could also be described as having “an osmolarity of 300”.If you could count every particle dissolved in the water of that compartment, you would see that in all the compartments there are the same number of particles: 300 million particles per liter, expressed as “300 million osmoles” or “300 mili-osmoles”.Therefore, size and composition (what particles are dissolved in the water) does not have an effect on the number of particles dissolved in the water of each compartment.Although the volume and substances dissolved in the fluid of one compartment is different than another compartment, each compartment has the same number of particles dissolved in the water.Body fluid compartments have different sizes and volumes, and different compositions.If you manipulate one body fluid compartment, it has an effect on another compartment.Transcellular fluid(the water enclosed in chambers lined by epithelial membranes) (1%) * * *.Plasma fluid(the water inside blood vessels, but not in blood cells) (9%).Interstitial fluid(the water immediately outside cells, between and around cells) (30%).Most medical solutions are calculated in units that don’t require a periodic table of elements, but if someone miscalculates a solution, and you inject it, and the patient crashes, you are just as liable, and you will be sued.Ĭompartments * Lumen of stomach These are stomach epithelial cells.Many of you will be applying IV care for patients, and sometimes doctors make mistakes, so you need to be able to catch these errors.Why do you need to understand body fluid compartments and osmolarity calculations?.Examples of osmotic pressure differences in body fluid compartments VII. Hydrostatic pressure- movement across capillaries V. Osmosis-movement across cell membranes due to unequal particles B. Review of Simple diffusion of solutes IV. Types of transport across the membrane for solutes-Protein transporters C.
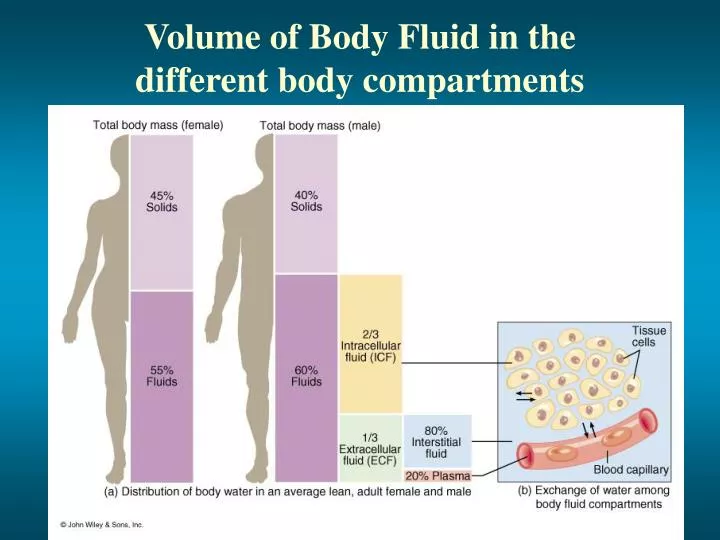

How do we have different composition/ movement of solutes A. Water compartments of the body A.Intracellular B.
#Fluid compartments free#
Body Fluid Compartments Free interactive Physio tools
